Quality Management Systems (QMS) are essential in each sector of the Food Industry, to ensure safe, quality food for the consumer. Food Safety and Quality audits are commonly used across the food industry to evaluate management systems, obtain certification from certain food and quality standards, assess the conditions of premises and products, and so on.
More and more Food Processing Plants are adopting Electronic Quality Management Systems to enhance their competitiveness on the global market and in response to growing pressure for consistent product quality, as well as the need for companies to demonstrate their quality management practices in order to meet the requirements of both legislation and the quality assurance practices of their customers.
After two years of continuous development and testing with significant input and validation from some of our largest food processing customer, Emydex in April 2018 released their Version 5 Electronic Quality Management System for Meat, Fish and Food Processors. The new Emydex EQMS stores all quality data in one centralised location, whether hosted in the cloud or installed locally on premise, depending on customer preferences.
The system provides Quality and Technical managers with accurate and holistic view of all Quality metrics and performance data as it happens in real-time across the organisation.
One core feature of the system is the ability to build and run in-house configured Quality checks and audits (workflows). By ‘in-house’ we mean QC workflows designed and built by QC staff, not the organisation’s in-house IT department. QC workflows are a series of Quality checks, questions and processes all wrapped into one.
The workflow builder offers QC users the ability to add conditional pathways, whereby the following questions presented are based on previous answers received from the operator i.e., if answer is A then 1, if B then 2. It also offers the ability to raise and track completion of follow up actions arising during audits and quality checks via email notifications. Any quality workflow in the system can be allowed to run at any time or can operate to a pre-defined schedule e.g., a pre-operation check, post-operation check or interval production check every two hours.
There are a number of major benefits arising from creating and assigning pre-defined schedules to QC workflows, including allowing management to identify when a QC workflow was:
- Missed
- Not started or completed on-time
- Completed in a time much faster than the average taken by other QC users, indicating that perhaps the survey was completed without adequate care and attention.
There are no hiding places in summary. The system also provides users with the option to indicate the reason why the check was missed, and automatically triggers follow up actions.
The Emydex EQMS provides the ability to create an electronic view (‘grid view’) for QC operators and managers to review and analyse QC data captured on the plant floor in detail. It also provides the option to generate in-house designed formatted reports that can be saved as a PDF, printed or sent to 3rd Parties, Auditors or emailed internally.
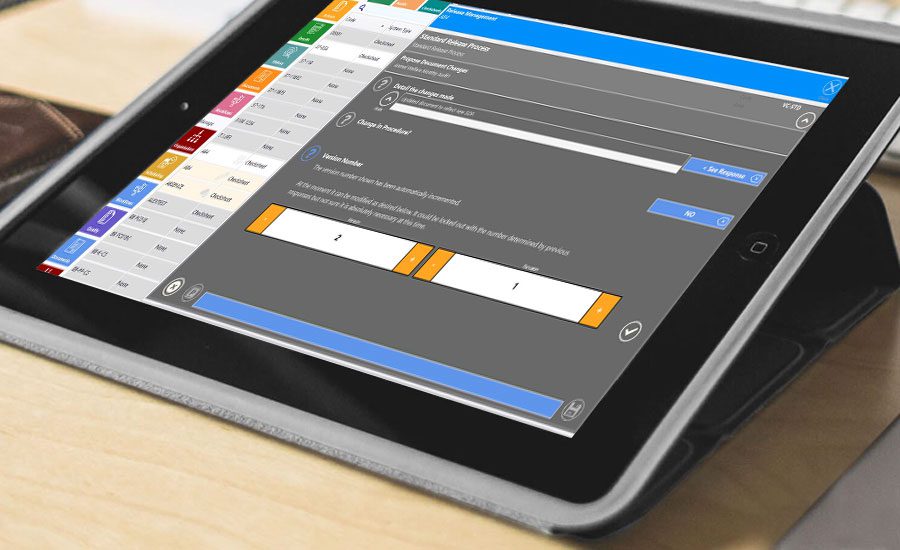
Additionally the system includes a fully functional document management and version control system, meaning documents can be uploaded to the EQMS system and readily made available to users as they complete workflows eg. retrieving details of a product spec during a product check and comparing the product on the line to the image in the documented product specification.
All documents and workflows can be managed via version control. This allows the back-office users to ‘check-out’ documents and workflows to make changes without affecting the Quality operation on the floor. Once they are happy with the changes, they can submit the document or workflow for approval and once approved, it becomes the active version on the floor.
For any Food processors that have already implemented the Emydex Production control and Traceability reporting modules the factory floor, a major advantage of the Electronic Quality Management System, can run the EQMS in tandem with their Emydex production system giving the ability scan product bar-codes, retrieve traceability details from Emydex’s production system database, and perform validation from the Electronic Quality Management System against production data stored in their Emydex Production system, something not possible with independent 3rd party QC systems.
The system incorporates both photographic as well as video evidence data capture.
To date the EQMS system has been implemented in numerous meat, fish and food processing plants, with both existing as well as new Emydex customers in Ireland, UK, Namibia and Australia with processors including Dawn Meats Group, Kepak Group, MeatCo Namibia as well as Dawnfresh Seafoods in Scotland. Dawnfresh currently operate the system on over 30 factory floor QC tablets spread over their two production facilities in Uddingston and Arbroath in Scotland.
A summary of some of the main features included in the system is below. If you are interested in an on-line demonstration or running a one-month free trial, please contact Shane Hayes, Emydex Technical Sales Manager on +353 1 8855990 or email shane.hayes@emydex.com
EMYDEX QUALITY MANAGEMENT SYSTEM DEMO VIDEO
Summary features include:
Production Calendars and Schedules
- Create calendars to keep track of scheduling
- Define operating hours/days
- Specify hourly, daily, weekly & monthly workflows
- Checks to be completed pre, post or during production
- Missed workflow notifications – automatically notify when a workflow is missing or not completed
Schedule Workflows
- Schedule Workflows with a click of a button. Apply zero, one or many schedules to a workflow as required
Roles & Responsibilities
- Assign QC Workflows only to those responsible for them.
- Assign Actions only to those with authority to complete them.
- Email notifications for assigned actions.
CAPA + RCA + PM Faults
- Construct your own Corrective/Preventative Action path to ensure you collate all data against the record that was non-conforming
Document Management Storage
- Upload, store and maintain your documents within the system. access these documents during your workflows on the production floor
- Procedures documents can be shared / accessed across multiple sites / plants
Version and Revision Control
- Build your various version and revision control for all documents and workflows. Know who changed what, when and why.
Organisational Set-Up
- Setup the company’s organisational structure within the system to control user access.